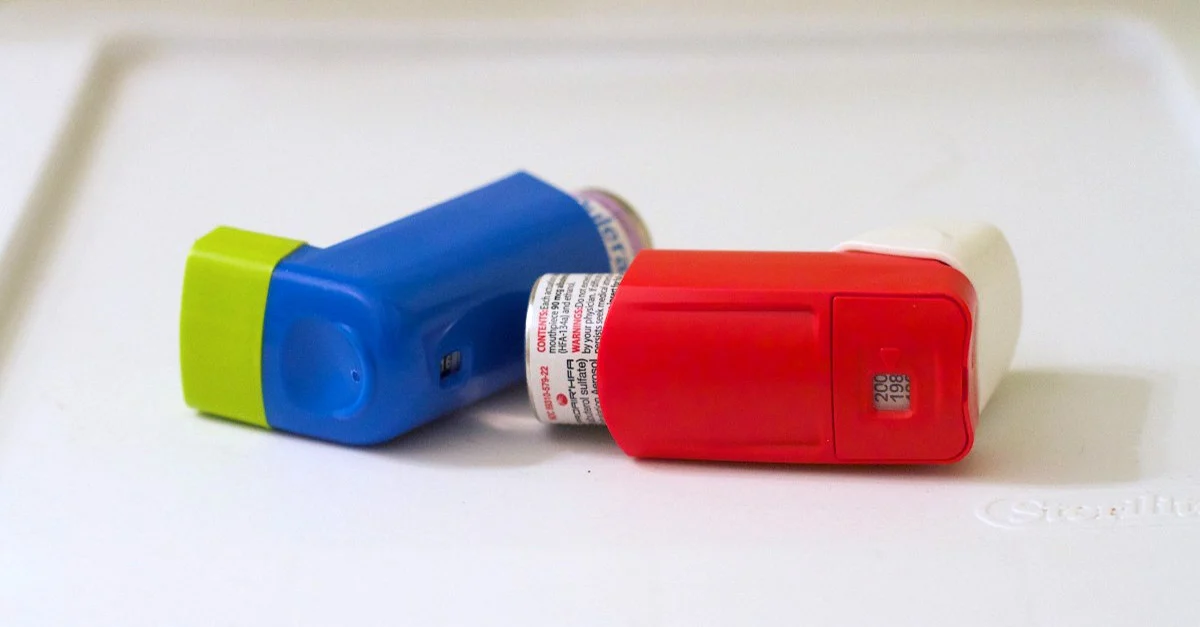
Brand name inhaler manufacturers tinker with patents and use gimmicks the Food and Drug Administration (FDA) shouldn’t allow to thwart the availability of low-cost generic inhalers. And profits are so high that a tobacco manufacturer took over an asthma inhaler company to “diversity its portfolio.”
A research article in Health Affairs reveals that manufacturers recycle the same patents on multiple inhalers from different classes and shift old ingredients to new devices. Of the 62 inhalers for asthma and chronic obstructive pulmonary disease (COPD) approved by the FDA during the past 35 years, only one contained an active ingredient with a new mechanism of action. More than half of the patents issued were for the actual inhaler devices.
The researchers conclude that regulatory and patent reform is critical to ensure that patent protection bestowed on brand name inhaler manufacturers better reflects the true clinical benefits of new products. Until then, patients will continue to experience the dual medical and financial burdens of asthma and COPD.